Popular Reads
Top Results
Can't find what you're looking for?
View all search resultsPopular Reads
Top Results
Can't find what you're looking for?
View all search resultsIn search of halal vaccines and access to medicines
Change text size
Gift Premium Articles
to Anyone
I
ndonesia will host the first ever meeting of the heads of National Medicines Regulatory Authorities (NMRA) from about 56 members of the Organization of Islamic Cooperation (OIC) in Jakarta on Nov. 21 and 22, which will also be attended by delegates from the World Health Organization, UNICEF, the Islamic Development Bank and leading vaccines and medicine manufacturers.
They will discuss strategic issues of common concern impacting the Islamic world, including how to strengthen collaboration toward self-reliance in medicines and vaccines.
This meeting could not be timelier. Needless to stress the importance of the provision and access to safe, good quality and efficacious medicines and vaccines. Particularly given the fact that many OIC member countries are confronted with public health challenges, crises and emergencies, caused by many factors ranging from poverty to violence and armed conflicts. This is true in the case of 21 OIC countries, which are still the least developed (LDC).
A 2017 OIC health report published by the Statistical, Economic and Social Research and Training Center for Islamic Countries (SESRIC) presents worrying heath situations, particularly in countries affected by conflicts and still suffering from extreme poverty — if not immediately resolved through collective actions by countries in the Islamic world, it will adversely affect the ability of regulators in the countries to provide access to basic health, essential medicines and vaccines.
Subsequently, medicines regulatory authorities of the OIC countries are facing complex pressures and challenges. They must ensure their performance is up to international standards and be capable of providing a strong oversight role in order to guarantee that the medicines and vaccines marketed and consumed by the public are safe, of great quality and efficacious.
To this end, the Food and Drug Monitoring Agency (BPOM) has a strategic role to play to ensure that the issues of collaboration for self-reliance of medicines and vaccines will be high on the OIC’s agenda. OIC secretary-general Yousef Al-Othaimeen has also suggested that the BPOM take the lead in strengthening OIC cooperation to achieve self-reliance in and access to medicines and vaccines for the benefit of OIC
countries.
As the government’s regulatory authority responsible for drugs and food control, the BPOM has been recognized internationally as one of the functional NMRAs and as the authority of one of the countries that produces WHO-prequalified vaccines with a good export capacity.
BPOM’s membership since 2012 in the Pharmaceutical Inspection Cooperation Scheme (PICS) — an international cooperative arrangement between 52 regulatory authorities from around the world in the field of Good Manufacturing Practice (GMP) of medicinal products — also proves its good standing.
Such a recognition provides a positive contribution for Indonesia’s export competitiveness in the country’s effort to boost exports and market expansion, showing that Indonesia is capable of providing international markets with safe, efficacious and high quality vaccines, including to OIC member countries.
Access to and availability of safe, high quality and affordable drugs are very important in preventing and curing diseases as well as maintaining and improving health, especially in OIC countries that still have a high prevalence of various diseases.
At present, drug manufacturing capacities within OIC member countries are still low, making them dependent on imported medicines from non-OIC countries and other important sources of medicines such as UNICEF. Only a handful of OIC member countries possess drug- and vaccine-manufacturing capabilities, namely Indonesia, Iran, Senegal, Uzbekistan, Bangladesh, Tunisia and Egypt.
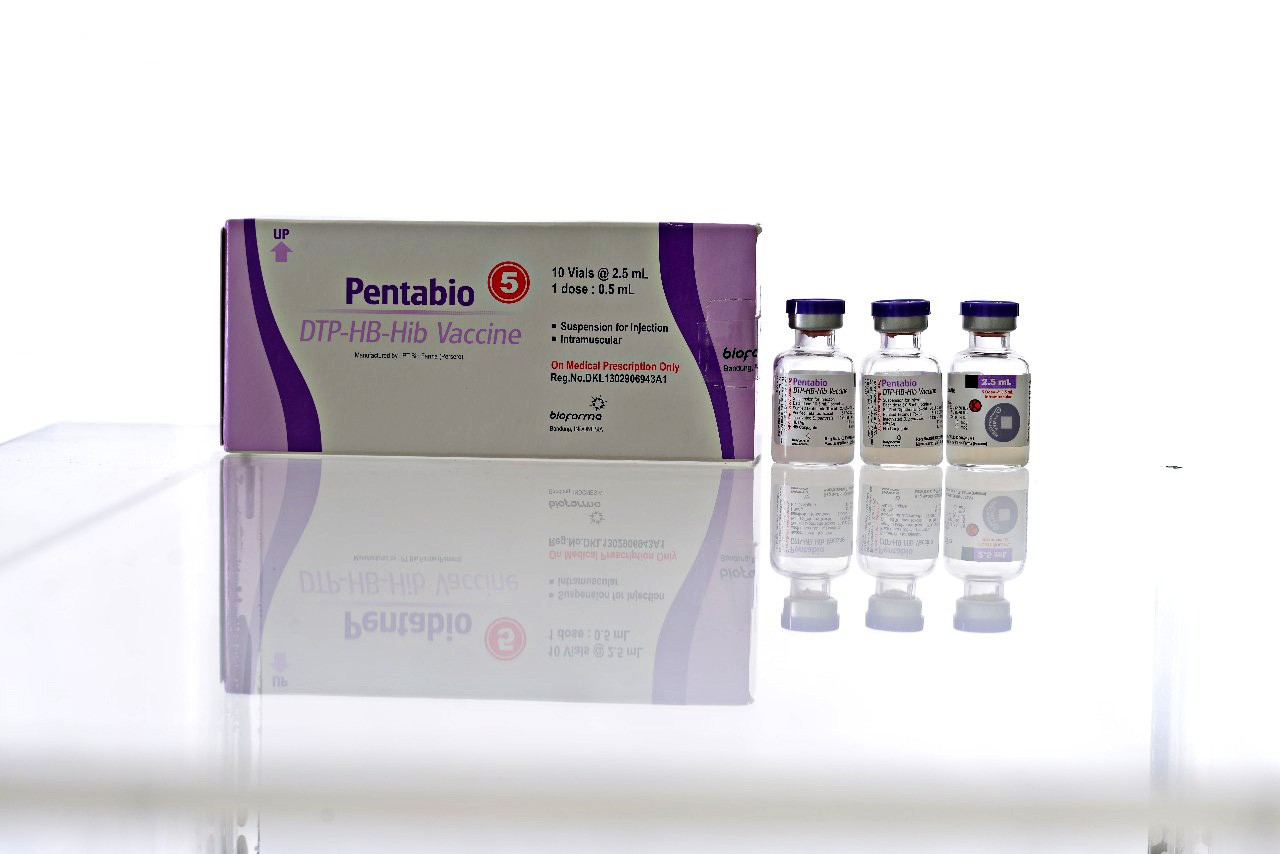
Vaccines produced by BioFarma, the state-owned vaccines producer of Indonesia, are used in more than 140 countries worldwide, mostly in developing countries, including in 49 out of the 56 OIC countries. UNICEF and other United Nations bodies have also used Bio-Farma vaccines in their programs.
Against this backdrop, this forthcoming meeting will identify and deliberate key issues that are highly relevant to experiences of OIC countries, especially from medicine regulatory authorities’ perspectives.
The meeting will discuss the current status of medicine regulation in OIC countries, the important role played by NMRAs in ensuring equitable access to safe, efficacious and quality medicines and the importance of developing and harmonizing standards of medicines and vaccines.
The need for halal medicines and vaccines is also a challenge for the Islamic world. Strategic solutions are needed, not only in supporting their production but also in making sure the vaccination program runs smoothly.
This forum offers opportunities to encourage the development of halal products so that they can expand the scope and increase the success of health programs in OIC member countries.
The delegates will also discuss the challenges faced by member countries in tackling substandard medicines and vaccines including counterfeit ones. OIC member countries will share their experiences in implementing strategies to address substandard and/or counterfeit medicines and vaccines, so that collaboration strategies/programs can be formulated to eradicate their circulation.
This timely forum is expected to produce a collaboration framework among NMRAs of OIC member countries to strengthen collaboration in the form of the Jakarta Declaration and Plan of Action to provide member states with a platform for continuous collective and concrete action, including through collaborations between pharmaceutical and vaccines manufacturers in OIC countries.
***
The writer is the head of the Food and Drug Monitoring Agency (BPOM). The views expressed are her own.