Popular Reads
Top Results
Can't find what you're looking for?
View all search resultsPopular Reads
Top Results
Can't find what you're looking for?
View all search resultsRegions may sanction COVID-19 anti-vaxxers amid plans for emergency use
Jakarta has issued a bylaw that stipulates sanctions for residents refusing to participate in the COVID-19 vaccination program.
Change text size
Gift Premium Articles
to Anyone
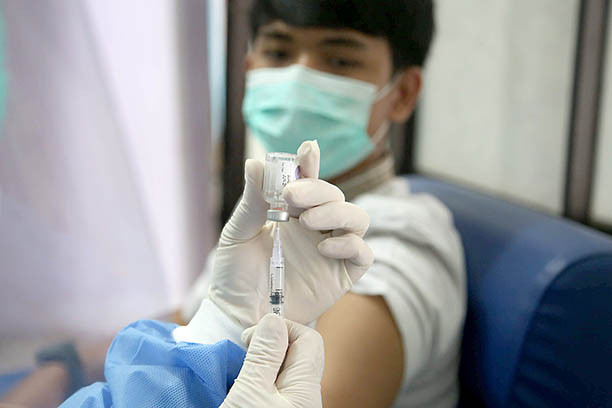 Test run: A medical professional at a community health center (Puskesmas) in Tapos, Depok, West Java, administers a vaccine during a COVID-19 vaccination simulation on Oct. 21. The medical simulation was attended by West Java Governor Ridwan Kamil. (JP/P.J.Leo)
Test run: A medical professional at a community health center (Puskesmas) in Tapos, Depok, West Java, administers a vaccine during a COVID-19 vaccination simulation on Oct. 21. The medical simulation was attended by West Java Governor Ridwan Kamil. (JP/P.J.Leo)
R
egional administrations are considering issuing bylaws as a legal avenue to sanction people who refuse to be vaccinated against COVID-19.
West Java Governor Ridwan Kamil said he had instructed his staff to review the legal aspects of such a bylaw.
“We are looking into the issue of whether people who refuse to get the COVID-19 vaccine will be harmful [to other] or if sanctioning them would violate their rights,” the governor said on Thursday as quoted by kompas.com.
He added that the West Java administration would intensify efforts to educate communities and raise awareness about the vaccine, as the central government was preparing to launch the first COVID-19 vaccination program in the country.
Read also: COVID-19 vaccine may be ready by year-end, says WHO's Tedros
The Jakarta administration issued on Monday a new bylaw on COVID-19 handling that also stipulates a sanctions for people resisting the vaccination program.
Article 30 of the bylaw stipulates that anyone deliberately refusing to undergo COVID-19 treatment and/or vaccination will be fined up to Rp 5 million (US$340), according to a copy of the bylaw’s final draft obtained by The Jakarta Post.
The possibility of sanctioning COVID-19 anti-vaxxers was raised as the central government plans to secure emergency-use authorization as soon as possible for several possible vaccines. The government hopes to secure authorization in November and roll out the vaccination program as early as January 2021.
However, the plan has been criticized by doctors across the country who urged the government not to rush the vaccination program and instead wait for the results of ongoing trials of possible vaccines to be published.
The Indonesian Doctor Association (IDI) said the government must choose vaccines that had proved effectiveness, safety and immunogenicity, or the ability to induce immune responses.
No vaccine has yet been approved for full use around the world. At least 12 possible vaccines are undergoing phase three of clinical trials, but none have passed the tests, the New York Times reported.
Read also: COVID-19 vaccines – A case for being cautiously optimistic?
Even so, Indonesia is pushing for emergency-use approval for three vaccines from Chinese companies Sinovac Biotech Ltd, China National Pharmaceutical Group (Sinopharm) and CanSino Bio. The government had sent a team that includes representatives of the Food and Drug Monitoring Agency (BPOM) to china to study data from the trials.
“They will proceed as planned,” said Achmad Yurianto, the Health Ministry’s then-director general for disease control and prevention.
On Friday, he was transferred from his director general post to become a Health Minister expert staffer.
Yurianto claimed the three possible vaccines had completed their trials in Brazil, Canada and Turkey. However, Reuters reported that Canada had ended its vaccine partnership with CanSino in August, while the Sinovac trials in Turkey had only started in September.
Brazilian researchers, meanwhile, found the Sinovac vaccine to be safe in a trial of 9,000 volunteers. However, the country would only release data on its effectiveness after the trials are completed on 13,000 volunteers.









