Popular Reads
Top Results
Can't find what you're looking for?
View all search resultsPopular Reads
Top Results
Can't find what you're looking for?
View all search resultsNo 'corners cut' to approve Pfizer-BioNTech vaccine: UK regulator
The Medicines and Healthcare products Regulatory Agency (MHRA) said it had used overlapping trials and "rolling reviews" since June to reach the determination in record time.
Change text size
Gift Premium Articles
to Anyone
B
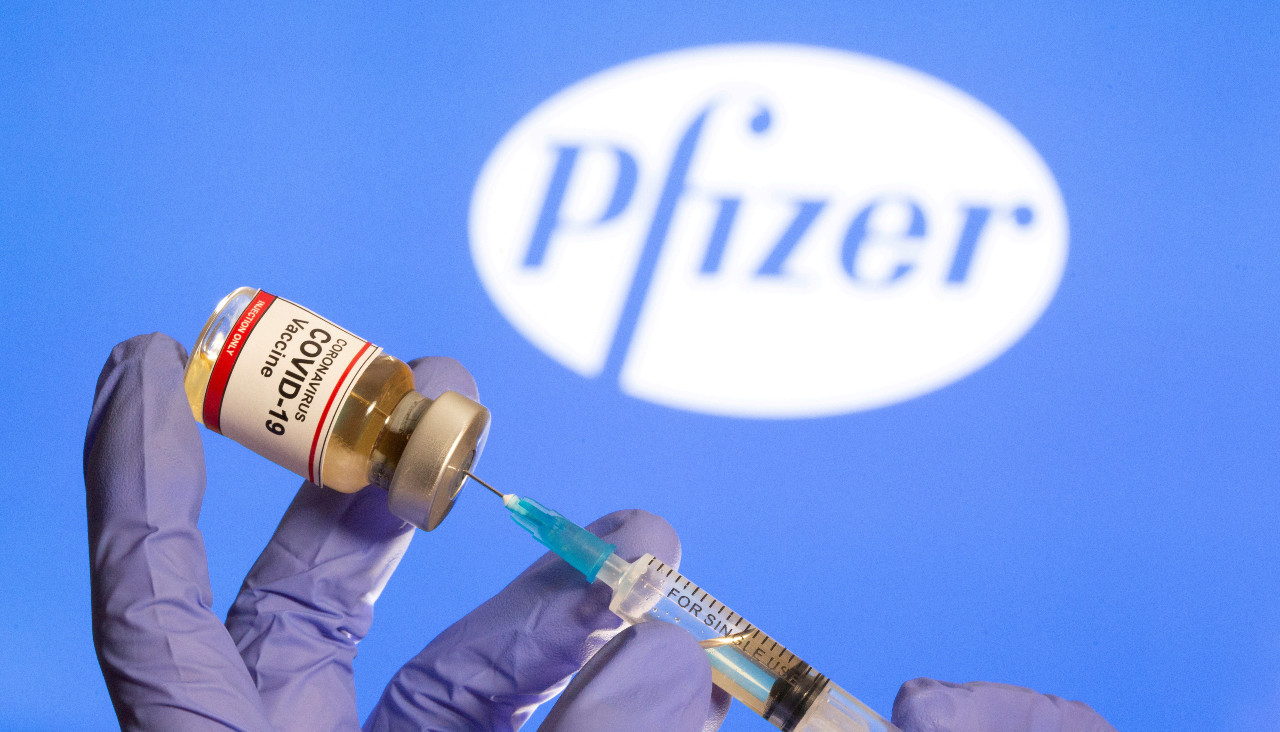
ritain's independent medicines regulator said on Wednesday no "corners have been cut" in its recommendation to approve Pfizer-BioNTech's COVID-19 vaccine for general use.
The Medicines and Healthcare products Regulatory Agency (MHRA) said it had used overlapping trials and "rolling reviews" since June to reach the determination in record time.
"That doesn't mean any corners have been cut –- none at all," June Raine, its chief executive, said at a press conference. "The safety of the public will always come first."
Wednesday's announcement saw Britain become the first Western country to approve a COVID-19 vaccine for general use, with jabs set to begin for the most vulnerable from next week.
The Pfizer-BioNTech vaccine is one of several expected to gain regulatory approval in the coming weeks as similar reviews are completed.
Read also: No emergency vaccine approval this year in Indonesia, says BPOM
Raine said the MHRA's "rigorous assessments" of the jab began in June after interim results from early trials by US giant Pfizer and German newcomer BioNTech became available.
"If you climb a mountain, you prepare and prepare," she added.
"On 10th November we were at base camp. When we got the final analysis we were ready for that last sprint that takes us to today."
British Health Secretary Matt Hancock said earlier the country's state-run National Health Service will start inoculations with 800,000 doses "early next week".
That will be ramped up to "millions" of jabs by the end of the year.
The mass vaccination program will begin with elderly care home residents and frontline health and social care staff.