Popular Reads
Top Results
Can't find what you're looking for?
View all search resultsPopular Reads
Top Results
Can't find what you're looking for?
View all search resultsVaccine safety and effectiveness must be ensured: Jokowi
“Because if not – if there’s even one problem – it will create public distrust in the vaccination effort,” he said.
Change text size
Gift Premium Articles
to Anyone
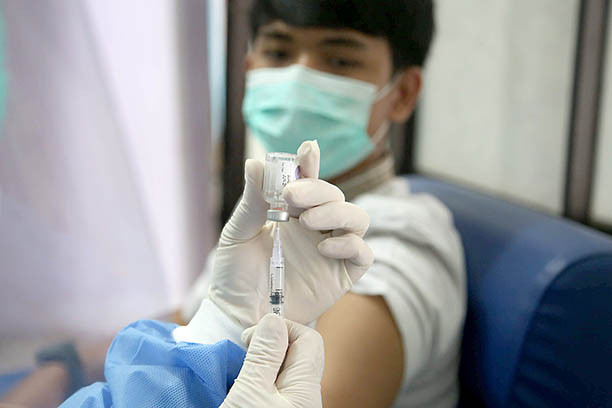 Test run: A medical worker at a community health center (Puskesmas) in Tapos, Depok, West Java, administers a vaccine during a COVID-19 vaccination simulation on Oct. 21. (JP/P.J.Leo)
Test run: A medical worker at a community health center (Puskesmas) in Tapos, Depok, West Java, administers a vaccine during a COVID-19 vaccination simulation on Oct. 21. (JP/P.J.Leo)
P
resident Joko “Jokowi” Widodo has instructed his Cabinet to ensure the safety and effectiveness of the COVID-19 vaccines to be used in the national vaccination program.
The two issues were the top concerns of experts and the public, he said.
“Safety means that all vaccines given to people must first go through a series of proper clinical trials,” the president said during a limited Cabinet meeting at Merdeka Palace on Monday. “Because if not – if there’s even one problem – it will create public distrust in the vaccination effort.”
Each stage of the trials, he said, must be based on scientific processes and data, as well as proper health standards. He warned his Cabinet not to let excessive haste jeopardize the testing process.
“Be careful, and don’t rush for a vaccine and overlook health standards,” he said.
Previously, the Indonesian Medical Association (IDI), the Indonesian Society of Internal Medicine (PAPDI) and several other associations warned the government not to rush into the COVID-19 vaccination program as the government pushed for emergency use authorization for several COVID-19 candidate vaccines still under development.
Experts said the government was betting on the success of unfinished vaccines to solve the COVID-19 crisis in the country.
Read also: Don’t rush COVID-19 vaccines, doctors tell govt eyeing emergency use
During the Cabinet meeting, Jokowi ordered the COVID-19 mitigation and national economic recovery team, led by State-Owned Enterprises (SOEs) Minister Erick Thohir, to establish a public communication strategy for the vaccination program.
“With the help of the Communications and Information Ministry, [the team] must comprehensively explain the benefits and the implementation of the vaccine to the public in order to avoid misinformation and the spreading of hoaxes by various media platforms,” Jokowi said during his speech.
He pointed out that the involvement of religious organizations such as the Indonesia Ulema Council (MUI) and the country’s largest Islamic organizations, Nahdlatul Ulama (NU) and Muhammadiyah, would be key to winning public trust because of their role in the dissemination of information and halal certifications.
While some vaccines would be free of charge, Jokowi said, independently purchased vaccinations would have a price.
“I have asked for them to be offered at affordable prices,” the President said.
“I demand that a detailed timeline be finished soon. [...] When will the vaccination be implemented in regions? Who will get free vaccinations and who will pay for them? All of these things must be planned and prepared for in detail,” Jokowi told his ministers.
State-owned pharmaceutical holding company PT Bio Farma, in partnership with China’s Sinovac Biotech Ltd., is conducting phase three clinical trials on a candidate vaccine in West Java. The trials are expected to end in January.
The country has also secured millions of doses of potential COVID-19 vaccines produced by Chinese firms Cansino and Sinopharm, Emirati artificial intelligence Group 42 (G42) Health Care and British pharmaceutical company AstraZeneca.









