Popular Reads
Top Results
Can't find what you're looking for?
View all search resultsPopular Reads
Top Results
Can't find what you're looking for?
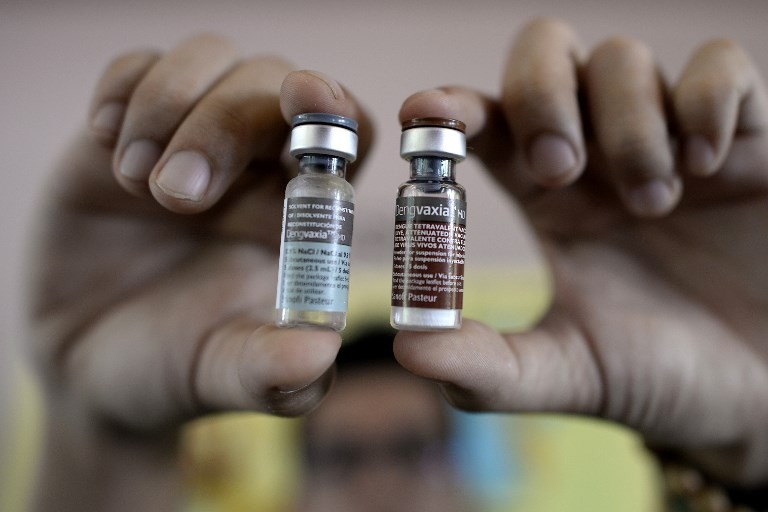
View all search resultsSanofi to reimburse Philippines for unused dengue vaccine
Change text size
Gift Premium Articles
to Anyone
F
rench pharmaceutical giant Sanofi is to reimburse the Philippine government for leftover doses of an anti-dengue vaccine whose use was suspended due to health concerns, the two parties said Monday.
The manufacturer said the refund had nothing to do with safety issues and was meant to improve ties with the Philippine health department, which is investigating the deaths of more than a dozen children injected with Dengvaxia.
Philippine regulators stopped the sale and distribution of the drug last month after Sanofi warned the shots could worsen symptoms for vaccinated people who contracted the disease for the first time.
The health department said in a statement it had issued a "demand letter" to the company's vaccine unit Sanofi Pasteur to refund 1.4 billion pesos ($27.8 million) for unused supplies of the drug.
"Our decision to reimburse for unused doses is not related to any safety or quality issue with Dengvaxia," said a statement from Sanofi Pasteur, which did not disclose the agreed amount.
"Rather Sanofi Pasteur hopes that this decision will allow us to be able to work more openly and constructively with the (health department) to address the negative tone towards the dengue vaccine in the Philippines today."
Philippine authorities have been investigating the deaths of 14 children who were among more than 830,000 given Dengvaxia last year in the world's first public immunisation programme against dengue.
After the programme began, Sanofi Pasteur released findings in November of a new study that showed Dengvaxia could lead to severe infections in some cases.
The disclosure triggered a public furore, with some parents blaming the vaccine for their children's deaths and a number of legislators accusing the government of endangering public health.
Health Secretary Francisco Duque has also threatened to sue the company if it had withheld crucial information on the vaccine.
Sanofi Pasteur has maintained that no death has been found to have been caused by Dengvaxia.
"Sanofi Pasteur strongly believes that this tone is due to a misunderstanding of the benefits and risks associated with the dengue vaccine," its statement said.
It cited "a lack of awareness amongst the general public, particularly parents of vaccinated children, that the overall benefit of dengue vaccination remains positive in high endemic countries like the Philippines".