Popular Reads
Top Results
Can't find what you're looking for?
View all search resultsPopular Reads
Top Results
Can't find what you're looking for?
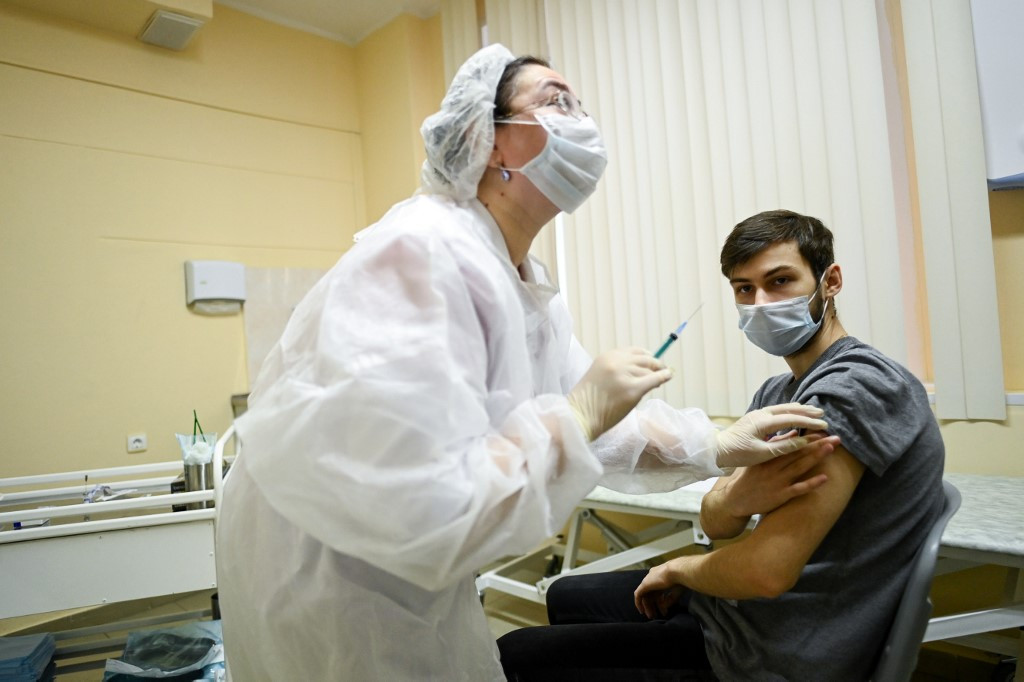
View all search resultsMexico, Germany warm to Russia's Sputnik V virus vaccine
The move is a boost to the Latin American nation's efforts to keep its immunization program on track in the face of limited supplies from other manufacturers.
Change text size
Gift Premium Articles
to Anyone
M
exico on Tuesday approved Russia's Sputnik V coronavirus vaccine for emergency use in the country, one of the worst hit by the pandemic, following the release of positive trial results.
The move is a boost to the Latin American nation's efforts to keep its immunization program on track in the face of limited supplies from other manufacturers.
Regulatory agency Cofepris "has just granted authorization for the emergency use of the Sputnik V vaccine," deputy health minister Hugo Lopez-Gatell told a news conference.
The shot -- named after the Soviet-era satellite -- faced criticism last year when it was approved in Russia before large-scale clinical trials.
But analysis of data from 20,000 Phase 3 trial participants, published Tuesday in medical journal The Lancet, suggests that the two-dose vaccination offers more than 90 percent efficacy against symptomatic Covid-19.
Mexican President Andres Manuel Lopez Obrador said last week after talking to President Vladimir Putin by telephone that Russia had agreed to provide 24 million doses of Sputnik V.
Mexico has more than 1.8 million known coronavirus cases and nearly 160,000 deaths -- the world's third-highest fatality toll after the United States and Brazil.
The country began mass immunization on December 24 but so far has only used the vaccine developed by US drugs giant Pfizer and its German partner BioNTech.
It has also authorized the shot developed by AstraZeneca and the University of Oxford, and has a deal to produce the vaccine in collaboration with Argentina.
Sputnik V is registered in more than a dozen other countries including former Soviet republics as well as allies such as Venezuela and Iran, as well as South Korea, Argentina, Algeria, Tunisia and Pakistan.
Meanwhile, German Chancellor Angela Merkel on Tuesday said all coronavirus vaccines "are welcome" in the EU once they have received regulatory approval, after Russia's Sputnik V jab was found to be 91.6 percent effective in trial results.
"Every vaccine is welcome in the European Union," Merkel said in an interview with German broadcaster ARD, as the bloc faces growing anger over a sluggish start to its inoculation campaign.
The veteran leader stressed however that all jabs must first be authorised by the European Medicines Agency (EMA), whose decisions can only be reached with "the necessary data".
"Today we have read good data for the Russian vaccine too," Merkel said.
She repeated that she had recently spoken to Russian President Vladimir Putin about the Sputnik vaccine, a conversation that fuelled hope another jab could be added to speed up efforts to end the pandemic.
Merkel said on January 21 that Germany had offered to assist Russia with its application process at the EMA, through Germany's Paul Ehrlich Institute.
"And if this vaccine is approved by the EMA, then we can talk about joint production or also about usage," Merkel said at the time.
This Handout photo made available by the German government's press office shows German Chancellor Angela Merkel speaking with French President Emmanuel Macron, via video link, at the Chancellery in Berlin, Germany, on May 18, 2020 on the effects of the novel coronavirus COVID-19 pandemic. (Bundesregierung/AFP/Sandra Steins)Sputnik V -- named after the Soviet-era satellite -- was approved in Russia months before results from its final-stage clinical trials were published, leading to scepticism from experts.
But the new analysis of data from 20,000 Phase 3 trail participants, published in The Lancet medical journal, suggests that the two-dose vaccination offers more than 90 percent efficacy against symptomatic Covid-19.
The encouraging results come at a time when the bloc's vaccine drive is dogged by supply bottlenecks and delivery delays.
In the ARD interview, Merkel again said that although the United States, Israel and Britain were further along with their vaccinations, Germany had been right to opt for a more cautious EU-wide approach.
While supply of the BioNTech/Pfizer and Moderna jabs will remain tight in the first quarter of 2021, Merkel insisted the pace would pick up later in the year, once production issues are resolved and more vaccine candidates are approved.
The outgoing chancellor renewed her promise that Germany was on track to offer every German a Covid-19 jab by late September.
But she also urged Germans "to hold their nerve a little while longer" and keep complying with tough curbs on public life, set to stay in place until at least mid-February.
"We still have a long way to go, and the hardest stretch is this winter," Merkel said.