Popular Reads
Top Results
Can't find what you're looking for?
View all search resultsPopular Reads
Top Results
Can't find what you're looking for?
View all search resultsCOVID-19-drugs
Ignoring outrage, Trump makes good on WHO pullout
President Donald Trump on Tuesday formally started the withdrawal of the United States from the World Health Organization, making good on threats to deprive the UN body of its top funding source over its response to the coronavirus.
EU looking to ramp-up production of COVID-19 antiviral remdesivir
The European Commission, the EU executive arm, said on Monday it was discussing ways of ramping up the production capacity of Gilead Sciences Inc, the US company that produces remdesivir, the only drug granted a conditional marketing authorization by the EU for its use in COVID-19 patients.
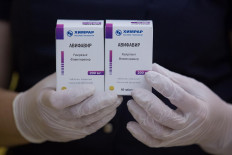
Russian fund steps up production of anti-viral drug approved by Moscow for COVID-19
Russia's sovereign wealth fund said on Thursday it will step up the production of the anti-viral drug Avifavir, an anti-influenza medicine which the Russian government has granted preliminary approval for treatment of COVID-19 patients.
World takes stock of COVID-19 drug remdesivir after US snaps up supplies
Remdesivir is in high demand after the intravenously-administered medicine helped shorten hospital recovery times in a clinical trial. It is believed to be most effective in treating COVID-19 patients earlier in the course of disease than other therapies like the steroid dexamethasone.
S.Korea to start talks on COVID-19 drug remdesivir purchases in August
South Korea has started distributing stocks of the COVID-19 treatment remdesivir that have been donated by Gilead Sciences Inc and plans to begin talks to purchase more supplies in August, its disease control agency said on Wednesday.
Chinese military researchers move a new COVID vaccine candidate into human trial
A Chinese military research institute has been approved to test its second experimental coronavirus vaccine in humans, the eighth candidate in clinical trials for China as it emerges as a front-runner in the global fight against COVID-19.