Popular Reads
Top Results
Can't find what you're looking for?
View all search resultsPopular Reads
Top Results
Can't find what you're looking for?
View all search resultshuman-trial
CanSino's COVID-19 vaccine candidate approved for military use in China
China's military has received the greenlight to use a COVID-19 vaccine candidate developed by its research unit and CanSino Biologics after clinical trials proved it was safe and showed some efficacy, the company said on Monday.
Chinese firm says coronavirus vaccine candidate shows promise in human test
China National Biotec Group (CNBG) said on Sunday that early human test results for a coronavirus vaccine candidate suggested it could be safe and effective, the second vaccine candidate from the firm to show encouraging results in a clinical trial.
Chinese military researchers move a new COVID vaccine candidate into human trial
A Chinese military research institute has been approved to test its second experimental coronavirus vaccine in humans, the eighth candidate in clinical trials for China as it emerges as a front-runner in the global fight against COVID-19.
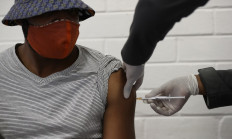
South Africa rolls out continent's first trials for COVID-19 vaccine
Oxford University rolled out Africa's first human trials for a potential vaccine against the new coronavirus in South Africa on Wednesday, as cases continue to rise and concerns grow over potential access to life-saving treatments.
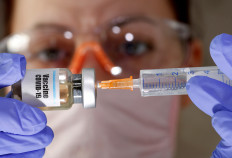
Chinese researchers launch phase-2 human test for possible coronavirus vaccine
Chinese researchers have started a second phase human trial of a possible coronavirus vaccine, the Institute of Medical Biology at Chinese Academy of Medical Sciences (IMBCAMS) said on Sunday, in efforts to further assess effectiveness and safety.
UK funds human trials of potential COVID-19 vaccine from Imperial College London
Scientists at Imperial College London will start the first clinical trials of a potential COVID-19 vaccine this week with more than 45 million pounds ($56.50 million) in backing from the UK government and philanthropic donors.
Indonesian COVID-19 vaccines to undergo strict human, animal testing
Indonesian researchers and authorities have assured that COVID-19 vaccine testing on animals and humans will meet ethical standards and undergo strict procedures, as animal rights organizations have objected to animal testing in formulating the vaccine.