Popular Reads
Top Results
Can't find what you're looking for?
View all search resultsPopular Reads
Top Results
Can't find what you're looking for?
View all search resultsSouth Africa rolls out second coronavirus vaccine trial
Change text size
Gift Premium Articles
to Anyone
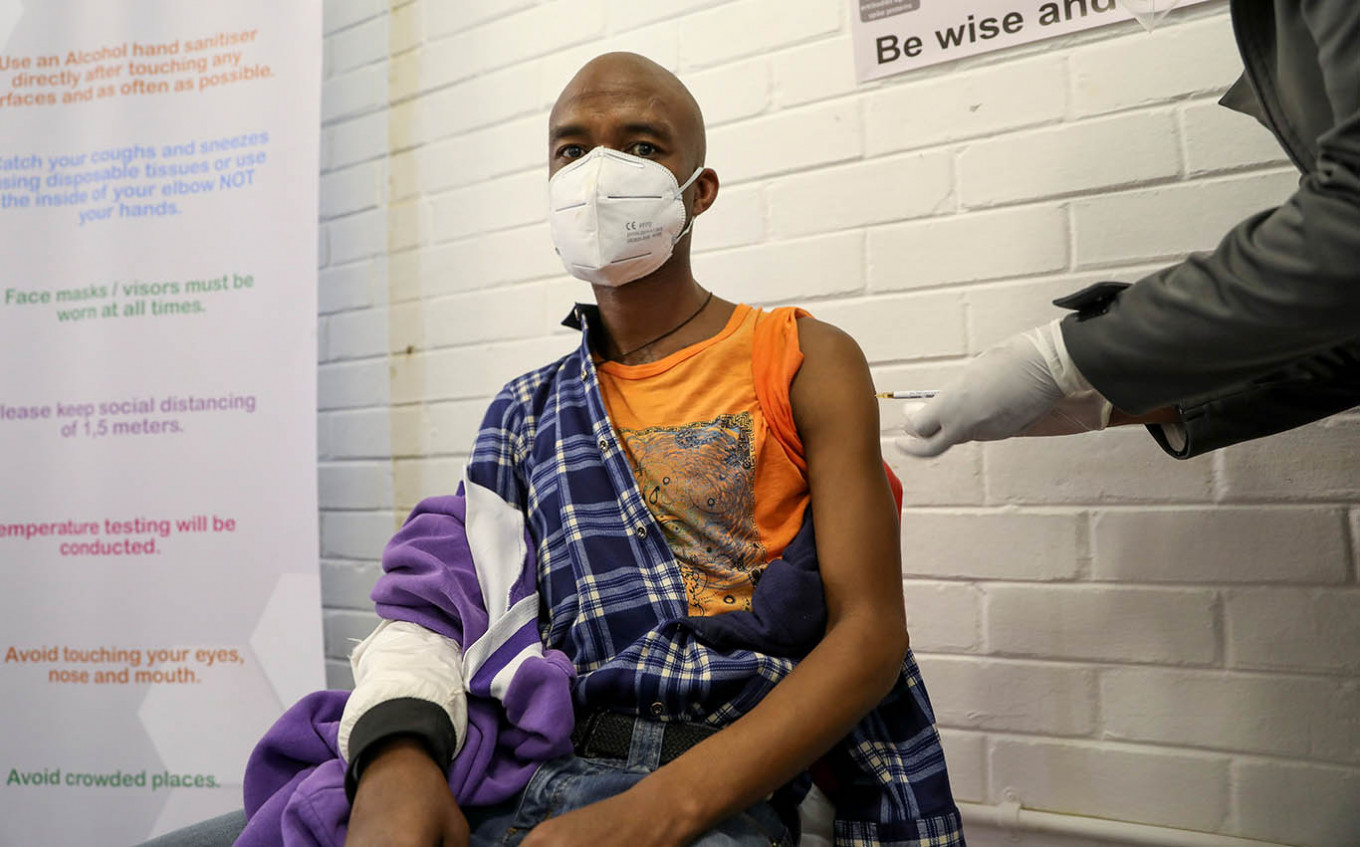 A volunteer receives an injection from a medical worker during the country's first human clinical trial for a potential vaccine against the novel coronavirus, at the Baragwanath hospital in Soweto, South Africa, June 24, 2020.South Africa will launch clinical trials of a US-developed coronavirus vaccine with 2,900 volunteers this week, the second such study in the African country worst hit by the disease, lead investigator Shabir Madhi said Tuesday. (REUTERS/Siphiwe Sibeko )
A volunteer receives an injection from a medical worker during the country's first human clinical trial for a potential vaccine against the novel coronavirus, at the Baragwanath hospital in Soweto, South Africa, June 24, 2020.South Africa will launch clinical trials of a US-developed coronavirus vaccine with 2,900 volunteers this week, the second such study in the African country worst hit by the disease, lead investigator Shabir Madhi said Tuesday. (REUTERS/Siphiwe Sibeko )
S
outh Africa will launch clinical trials of a US-developed coronavirus vaccine with 2,900 volunteers this week, the second such study in the African country worst hit by the disease, lead investigator Shabir Madhi said Tuesday.
Known as NVX-CoV2373, the vaccine was developed by US biotech company Novavax from the genetic sequence of SARS‑CoV‑2, the virus that causes the COVID-19 disease.
It will be administered to the first volunteer in the randomized, observer-blinded trial on Wednesday.
"It's a two-dose schedule, and they get two either vaccines or placebos... spaced three weeks apart," professor Madhi of the Johannesburg-based University of the Witwatersrand (Wits), told AFP.
With 589,886 cases and 11,982 deaths, South Africa is fifth in global rankings for countries with the most infections.
Since the country also accounts for 20 percent of global HIV infections according to UNAIDS 2020 data, 240 medically stable, HIV-positive adults will also participate in the clinical trial.
"It's critical that we understand how these vaccines work in populations that have HIV, especially in South Africa where they constitute up to about 12 to 15 percent of the adult population," said Madhi.
Wits University said studies of the Novavax vaccine in non-human primates have shown protection against the coronavirus infection in nasal passages as well as protection against lung disease.
Partly funded by a $15 million grant from the Bill & Melinda Gates Foundation, the trials are part of a larger study to be launched throughout the world to involve approximately 30,000 participants.
In June, South Africa piloted its first trial of a vaccine developed by the Britain-based Oxford Jenner Institute.
Some 2,000 people were injected with the vaccine known as ChAdOx1 nCoV-19 with 50 of the candidates having HIV.









