Popular Reads
Top Results
Can't find what you're looking for?
View all search resultsPopular Reads
Top Results
Can't find what you're looking for?
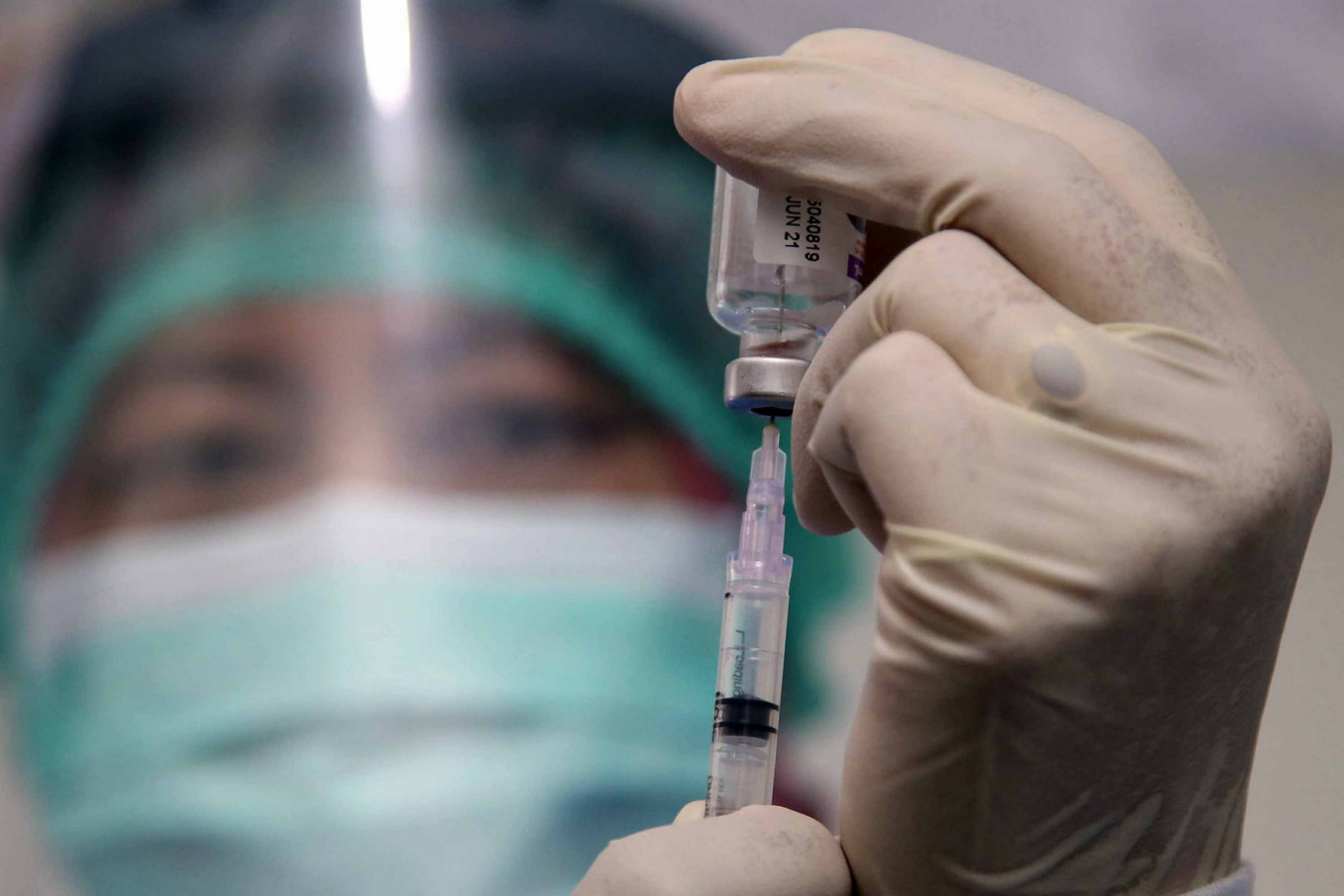
View all search resultsCOVID-19 vaccine 'key for herd immunity'
The most effective and safest vaccine was needed in a mass vaccination program aimed at reducing morbidity and mortality rates, said a KSP official.
Change text size
Gift Premium Articles
to Anyone
I
mmunization is the key to achieving herd immunity and curb the spread of COVID-19 once health experts successfully develop a COVID-19 vaccine, the Executive Office of the President (KSP) has said.
A principal expert at the KSP deputy II for human development, Brian Sri Prahastuti, said herd immunity could be achieved if the country succeeded in vaccinating at least 75 percent of the population.
“That way, 25 percent of the population who for some reason did not get the vaccine, will also benefit because virus transmission would be minimized,” Brian said in a statement on Friday.
The most effective and safest vaccine was needed in a mass vaccination program aimed at reducing morbidity and mortality rates, she added.
Brian said the government had been taking into account the principles of effectiveness and safety in developing a COVID-19 vaccine.
Indonesia has been actively involved in the clinical trials of China’s Sinovac vaccine through state-owned pharmaceutical holding company PT Bio Farma.
“We know that Bio Farma has high international credibility in vaccine production, so does Hasan Sadikin Hospital [RSHS] in vaccine clinical trials,” she said.
Read also: Road map for COVID-19 vaccination in final stage: Task force
RSHS is a public hospital in Bandung, West Java, which is conducting large-scale clinical trials of convalescent blood plasma therapy on COVID-19 patients, along with several other hospitals across Java.
Brian asked the public not to hesitate over the conclusion of the final trials because they involved the Technical Advisory Group on Immunization (ITAGI), and vaccine production would first gain emergency use authorization from the Food and Drug Supervisory Agency (BPOM).
The BPOM is currently visiting the Sinovac factory in China to assess the ongoing phase III clinical trials and the overall vaccine production process.
Bio Farma has submitted an interim report and will receive approval once the trials are completed, she said.
The company is currently running final-stage clinical trials of a vaccine developed by Sinovac, while Jakarta-based PT Kalbe Farma is working with South Korea’s Genexine on another possible vaccine.
The government has cooperated with various Islamic organizations to support the upcoming nationwide COVID-19 vaccination program, which it seeks to begin in November or December after uncertainties over the vaccine's halal status have been settled.
Vice President Ma’ruf Amin, who is the chair of the Indonesian Ulema Council (MUI), previously said the COVID-19 vaccine currently being prepared by the government did not have to be halal. (syk)
Editor’s note: This article is part of a public campaign by the COVID-19 task force to raise people’s awareness about the pandemic.