Popular Reads
Top Results
Can't find what you're looking for?
View all search resultsPopular Reads
Top Results
Can't find what you're looking for?
View all search resultsJava, Bali top priority to receive vaccines due to number of COVID-19 cases
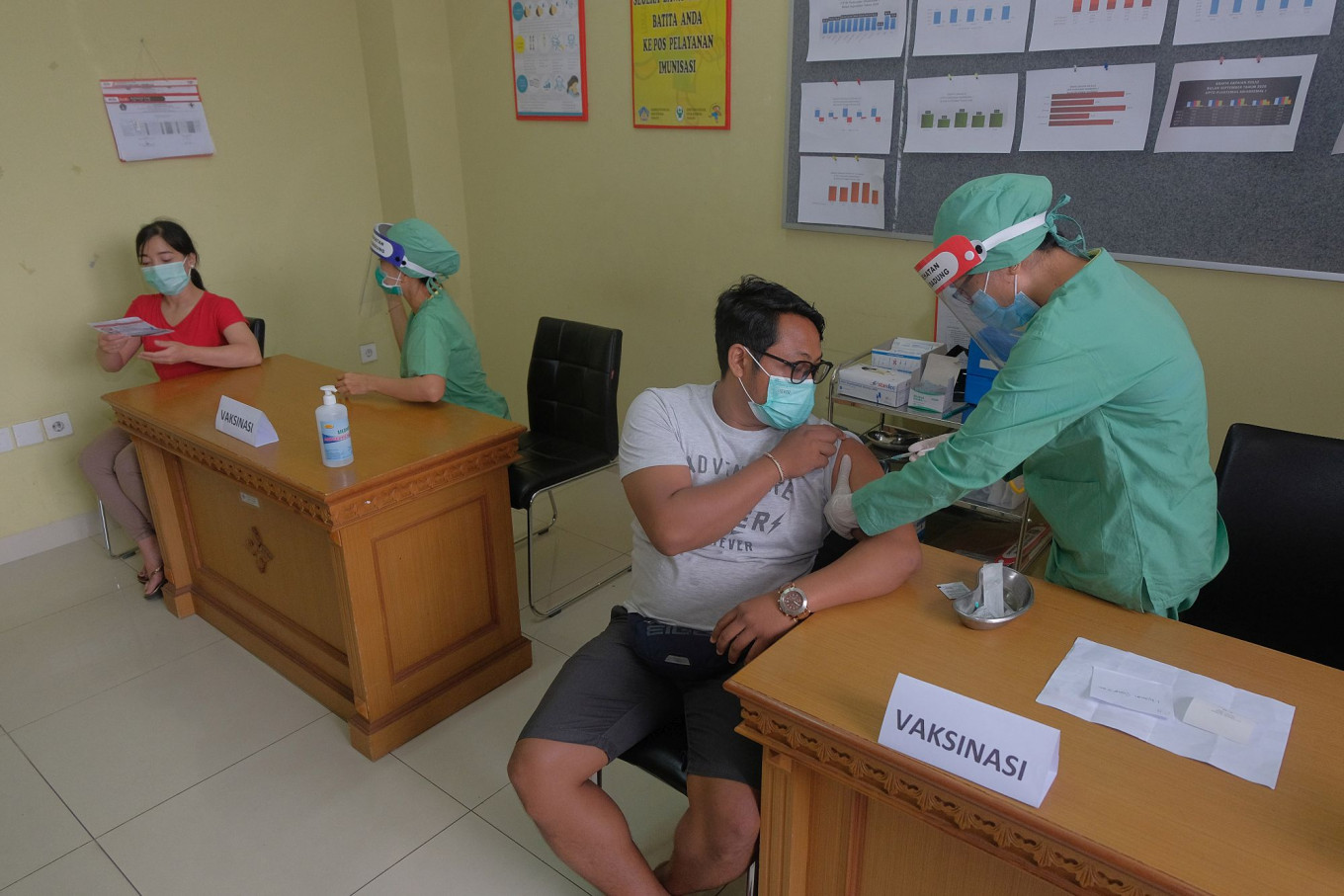
Coordinating Human Development and Culture Minister Muhadjir Effendy previously said President Joko “Jokowi” Widodo had ordered his administration to prioritize red zones in the vaccination plan.
Change text size
Gift Premium Articles
to Anyone
T
he government will prioritize Java and Bali islands for the first round of vaccination programs due to their high number of COVID-19 cases.
"Out of the 10 provinces with the most confirmed cases, most of them are in Java and Bali islands," national vaccination program spokeswoman Siti Nadia Tarmizi said on Wednesday as quoted by tempo.co.
According to National Disaster Mitigation Agency (BNPB) data on Dec. 9, active cases in seven provinces in Java and Bali had reached 50,892, totaling 376,112.
Central Java led the tally with more than 17,000 cases, followed by Jakarta with 11,700 known cases and West Java with more than 10,000 cases.
Siti Nadia said the high number active cases was triggered by dense populations in the said regions, adding that 60 percent of the country’s 267 million population resided in Java and Bali.
Read also: Bio Farma allocates 3 million doses of COVID-19 vaccine for health workers
National COVID-19 task force spokesman Wiku Adisasmita said administering vaccines to people in high-risk red zones was expected to curb virus transmission, adding that vaccination was not only aimed at saving a life but an entire population.
Therefore, Wiku reminded the public to limit their mobility while continuing to comply with strict health protocols prior to and after the national mass vaccination program.
“If the public neglects the health protocols, we will surely have case spikes,” he said, adding that the vaccinations would not guarantee an end to the pandemic.
Coordinating Human Development and Culture Minister Muhadjir Effendy previously said President Joko “Jokowi” Widodo had ordered his administration to prioritize red zones for vaccination.
“[We cannot] administer shots on random people but must instead [ prioritize] certain people based on risk zoning,” he said.
In the first phase of the national vaccination program in Java and Bali, the government is to utilize 1.2 million doses of a COVID-19 vaccine produced by China’s Sinovac Life Science Corp. Ltd., which arrived in Indonesia on Dec. 6. In total, Indonesia has secured 3 million doses of the Sinovac vaccine, in which the remaining 1.8 million doses are to reach the country in January.
Read also: Central Java, Bali, Papua need special attention: Task force
National economic recovery task force head Budi Gunadi Sadikin said Sinovac was set to send 45 doses of bulk vaccine for state-owned pharmaceutical company PT Bio Farma to process further. Out of 15 million doses of bulk vaccine set to arrive in Indonesia this month, Bio Farma will process them further to produce 12 million doses of ready-to-use vaccines.
“The remaining 30 million doses of bulk vaccine will reach [Indonesia] in January, and will be processed to become 24 million doses of final product vaccine in the next month,” said Budi.
Apart from Sinovac, the government will procure several other vaccines produced by AstraZaneca, China National Pharmaceutical Group Corporation (Sinopharm), Moderna, as well as Pfizer Inc. and BioNTech.
Bio Farma president director Honesti Basyri said he had also communicated with Pfizer and AstraZeneca for a possible future arrangement. Meanwhile, with Novavax, the company had signed a commitment for vaccine procurement.
“With Novavax, we have a commitment [to buy] 30 million doses, which we will further discuss,” he added.
Health Minister Terawan Agus Putranto said the mass vaccination program would be immediately conducted as soon as the Food and Drug Monitoring Agency issued emergency use authorization for the vaccines. (nkn)
Editor’s note: This article is part of a public campaign by the COVID-19 task force to raise people’s awareness about the pandemic.