Popular Reads
Top Results
Can't find what you're looking for?
View all search resultsPopular Reads
Top Results
Can't find what you're looking for?
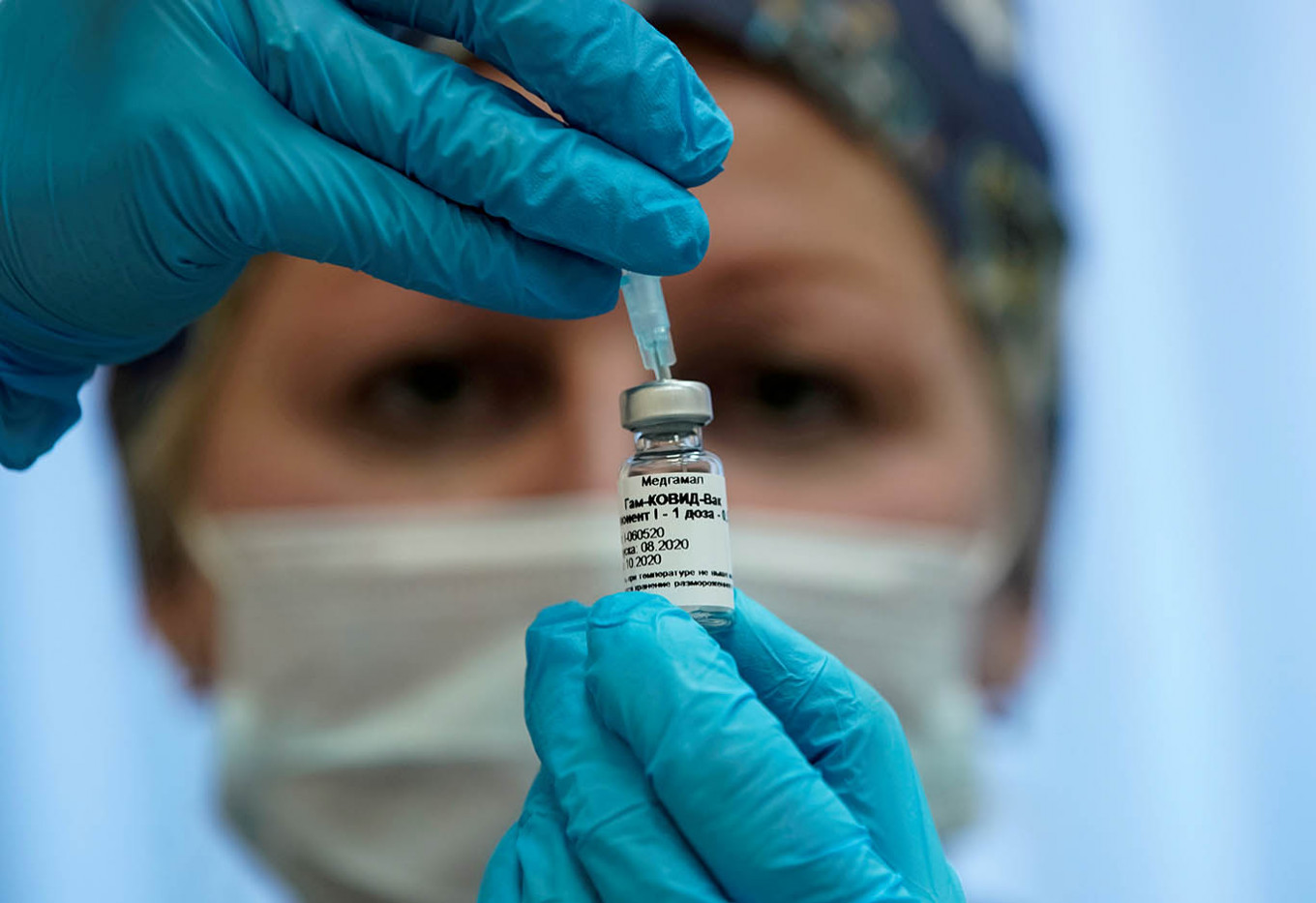
View all search resultsIndonesians welcome Sputnik V vaccine
Over 64 percent of Indonesian respondents in a survey conducted by global public opinion and data company YouGov have revealed that they wish to be vaccinated against COVID-19, while mentioning Russia among their top-three vaccine-manufacturing countries.
Change text size
Gift Premium Articles
to Anyone
Over 64 percent of Indonesian respondents in a survey conducted by global public opinion and data company YouGov have revealed that they wish to be vaccinated against COVID-19, while mentioning Russia among their top-three vaccine-manufacturing countries.
The same survey also showed that over 85 percent of Indonesian respondents prefer the human adenovirus vaccine platform used for the Sputnik V vaccine candidate over non-human adenovirus platforms.
According to the website for Sputnik V, sputnikvaccine.com, adenoviral vectors are considered extremely safe and are some of the easiest to engineer. Vectors are viruses that have had the gene responsible for replication removed. Therefore, they no longer pose any threat of infection. Scientists use vectors to transport genetic material from a different virus – one that is being vaccinated against – into a human cell.
As many as 12,000 people across 11 countries took part in the survey, which was aimed at studying opinions regarding potential COVID-19 vaccines. The respondents included people from Brazil, Vietnam, Egypt, India, Indonesia, Malaysia, Mexico, Nigeria, the United Arab Emirates, Saudi Arabia and the Philippines.
The survey, conducted from Oct. 9 to 19, also showed that 73 percent of total respondents were willing to be vaccinated against the coronavirus.
Meanwhile, four out of five respondents who were aware of the Sputnik V vaccine were willing to be vaccinated with it, increasing Russia’s confidence as a global vaccine manufacturer. The survey also revealed that over 30 percent of the respondents were aware of the vaccine.
“More than 2.5 billion people live in these countries, or over 30 percent of the world’s population. The survey became one of the largest on this topic and the first global survey involving the countries of the Middle East and Southeast Asia,” Russian Direct Investment Fund (RDIF) said in a statement.
Previously, the Russian government had contacted Indonesia’s Health Ministry over the possibility of Sputnik V vaccine distribution on Sept. 17.
“We have sent a letter to the ministry on Sept. 17. We’re still waiting for a response,” Russian Ambassador to Indonesia Lyudmila Vorobieva said on Oct. 15.
Read also: From US elections to COVID-19 vaccine, Russia responds
The vaccine, which is currently in phase three clinical trials, has shown 92 percent of effectiveness according to vaccine producer Gamaleya Research Institute. Forty-thousand people are participating in the trial, which commenced in Russia on Aug. 11.
The efficacy result itself was obtained through evaluating confirmed COVID-19 cases from 16,000 volunteers who had been injected with the vaccine candidate or a placebo. So far, no unexpected adverse events have been reported during the trial.
In September, the vaccine was administered to a group of volunteers who hailed from Russian hospitals in red-zone areas. Over 20,000 volunteers have been given the first shot as of Nov. 11, while over 16,000 volunteers have received the second shot of the vaccine. (dpk)